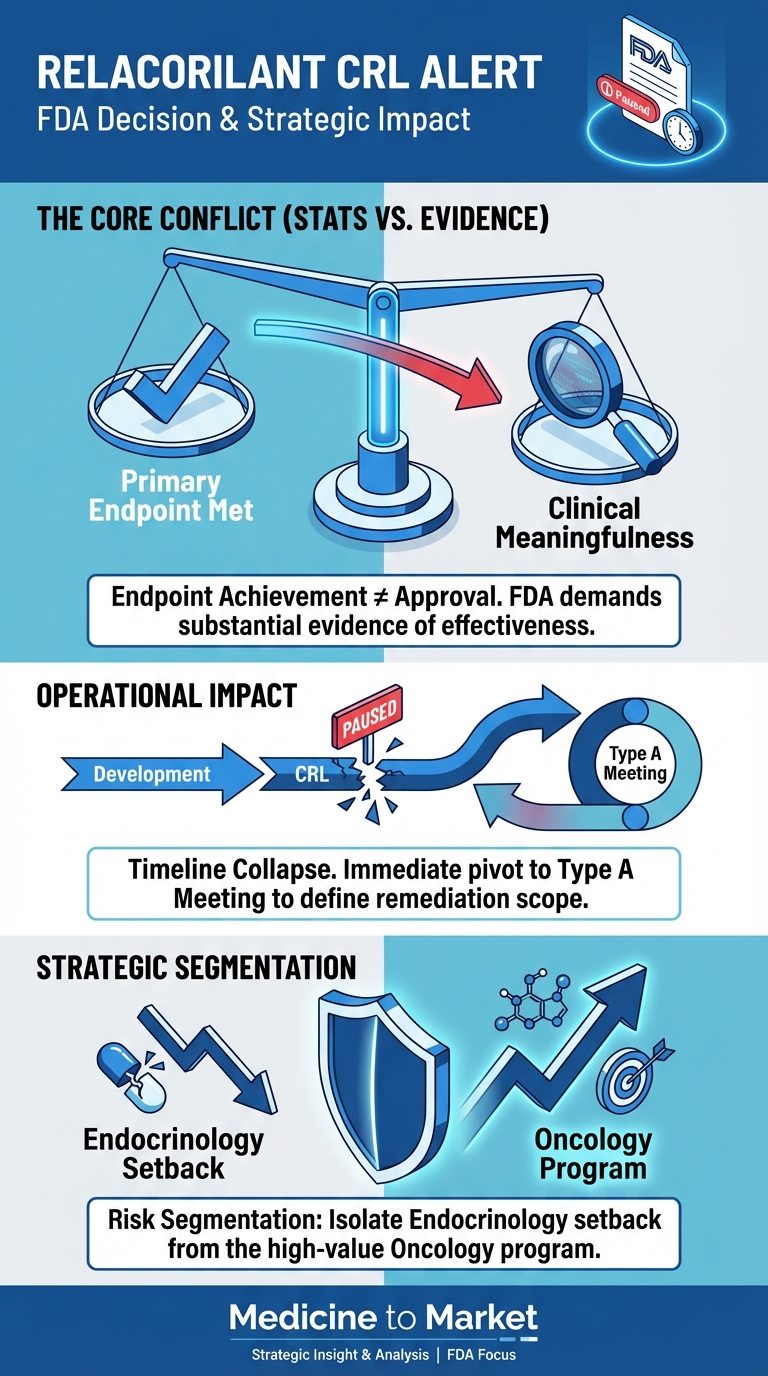
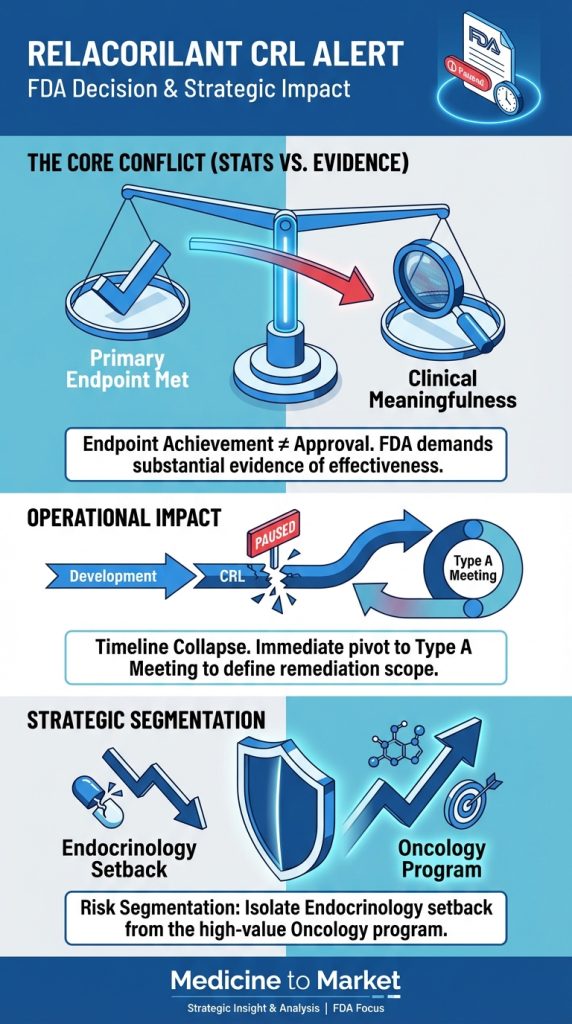
Boehringer Ingelheim has made a significant advancement in the treatment of fibrotic lung diseases with its PDE4 inhibitor, successfully completing two Phase 3 trials.
However, the mixed responses from Wall Street analysts have sparked conversations regarding the drug’s true efficacy and market viability.
With efficacy being described as ‘modest’ and concerns about its compatibility with other therapies, the future of this promising candidate in a competitive landscape remains uncertain.
This article delves into the recent trial outcomes, the analyst critiques, and the potential implications of these responses on the drug’s market prospects.

Key Takeaways
- Boehringer Ingelheim’s PDE4 inhibitor has passed two Phase 3 trials but faced mixed reactions from analysts.
- Concerns over the drug’s efficacy being described as ‘modest’ may limit its market potential.
- Analysts are wary of the drug’s effectiveness in real-world applications and its compatibility with other treatments.
Overview of Boehringer Ingelheim’s PDE4 Inhibitor and Phase 3 Trials
Boehringer Ingelheim has generated significant attention in the pharmaceutical industry with the recent announcement of its Phosphodiesterase 4 (PDE4) inhibitor, which has shown promising results in two Phase 3 trials focused on fibrotic lung diseases.
These trials are crucial as they represent the final stage of clinical testing before a potential market launch, providing substantial insights into the drug’s efficacy and safety.
Despite the positive results from the trials, Wall Street analysts have expressed mixed reactions, with many citing concerns over the candidate’s efficacy being described as ‘modest.’ This raises questions about how effective the drug will be in real-world applications, especially considering its limited potential for combination with existing therapies.
As the landscape for lung disease treatments becomes increasingly competitive, the mixed feedback from analysts reveals a cautious outlook regarding the drug’s market viability.
The results are crucial for Boehringer Ingelheim, which aims to address unmet medical needs in lung disease, yet the skepticism may affect investor confidence and the drug’s potential adoption by healthcare providers.
Analyst Concerns and Implications for Market Potential
The implications of the mixed feedback from Wall Street analysts extend beyond mere skepticism about the drug’s efficacy.
Investors are closely monitoring how Boehringer Ingelheim plans to navigate the competitive landscape for fibrotic lung disease treatments.
If the PDE4 inhibitor’s effectiveness remains unproven in broader patient populations, it could significantly hinder its adoption among healthcare providers who might prefer more robust alternatives.
Furthermore, the concern over its modest efficacy raises potential barriers to securing partnerships for combination therapy, which could limit the drug’s appeal in an increasingly crowded market.
As companies continue to innovate in lung disease therapies, Boehringer Ingelheim must demonstrate not only the clinical benefits of its PDE4 inhibitor but also articulate a compelling value proposition to gain traction with both investors and clinicians.