Adaptive Trial Design: When to Use It and When to Avoid It
Adaptive trial design — modifying a study’s protocol based on accumulating data — is one of the most discussed and
Summaries and analysis of industry press releases
Adaptive trial design — modifying a study’s protocol based on accumulating data — is one of the most discussed and
Every pharma brand plan has a KOL strategy. Most of them are built on the same foundation: identify high-prescribers and
Medical Science Liaisons occupy a structurally unusual position in pharma — they’re the scientific face of the company in the
Omnichannel is the most overused word in pharma commercial strategy. Every brand plan has an omnichannel section. Few brand teams
The clinical program is done. The PDUFA date is set. What happens next determines whether the asset reaches its commercial
Health economics and outcomes research is routinely treated as a specialty function sitting adjacent to commercial — the team that
The 340B drug pricing program started as a narrow safety-net mechanism and has evolved into one of the most contested
The first round of IRA drug price negotiation is complete. The results are a preview of the program’s operating logic
PBM formulary decisions are binary in their commercial impact: your drug is on the preferred tier with manageable patient cost-sharing,
The gap between a drug’s sticker price and what anyone actually pays is where most commercial strategy either gets built
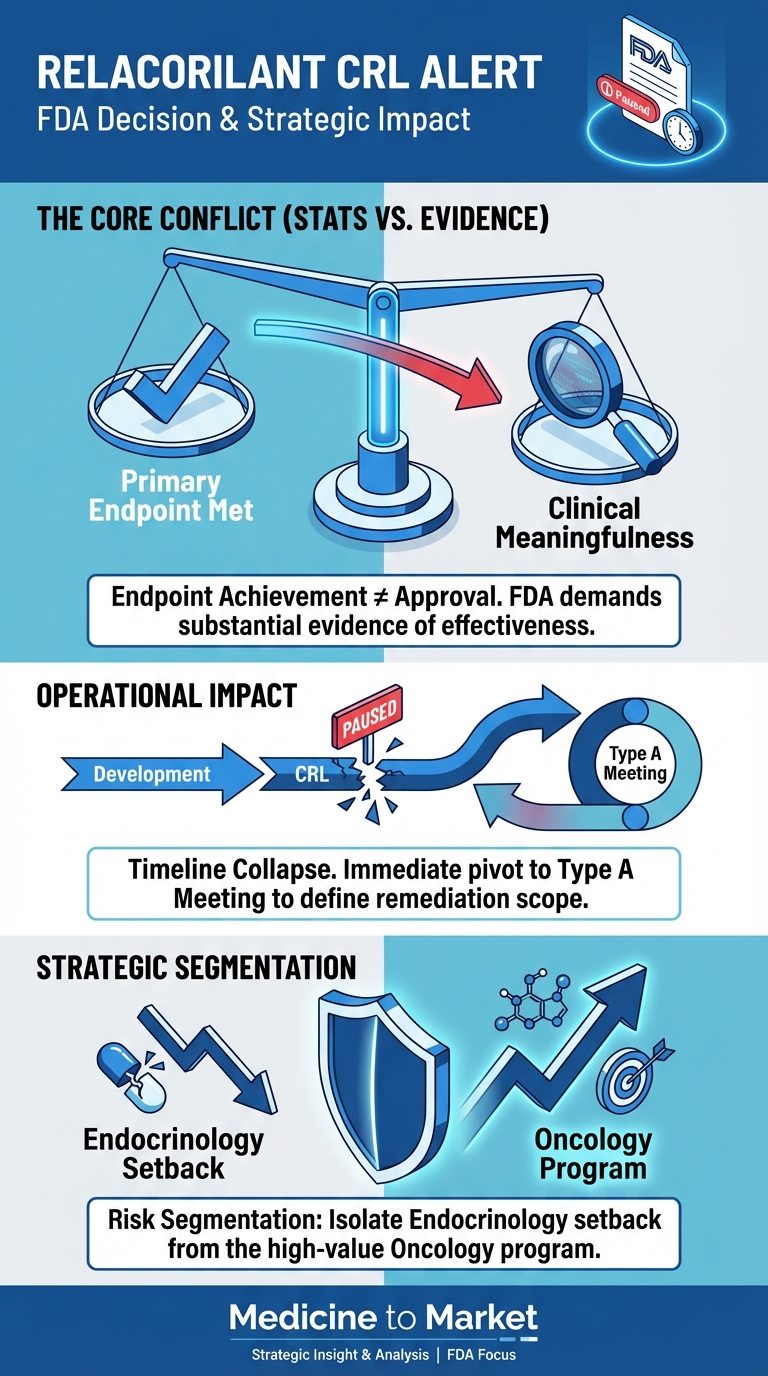
What’s Next for Corcept Therapeutics After FDA’s CRL for Relacorilant as a Treatment for Patients with Hypercortisolism Date: December 31,

In today’s fast pharma scene, brands act. Customer talk must hit, feel real. Channels mix—meet, talk, email, web. Pharma steers

Artificial intelligence (AI) shifts U.S. healthcare. AI touches care, tests, and drug find. AI screens images. AI spots new drugs.

In a significant shift aimed at enhancing its competitive edge in the biopharma industry, Novartis has announced a strategic workforce

The recent appointment of Richard Pazdur as the head of the FDA’s drug center has stirred up notable controversy and

The landscape of Alzheimer’s treatment took a notable downturn recently when Johnson & Johnson announced that its anti-tau monoclonal antibody

Navigating the complex landscape of pharmaceutical development requires a deep understanding of regulatory trends, market dynamics, and technological advances. In

In the intricate world of pharmaceutical regulation, the intersection of politics and science can greatly influence decision-making processes. Dr. Palak

November 21, 2025 HEADLINE FINDINGS: Alkermes escalates bidding war for Avadel Pharmaceuticals to $2.37 billion, intensifying competition for specialty CNS assets

In a significant turn of events, Ensoma, a Boston-based biotechnology startup dedicated to pioneering one-time genetic medicines, has announced drastic