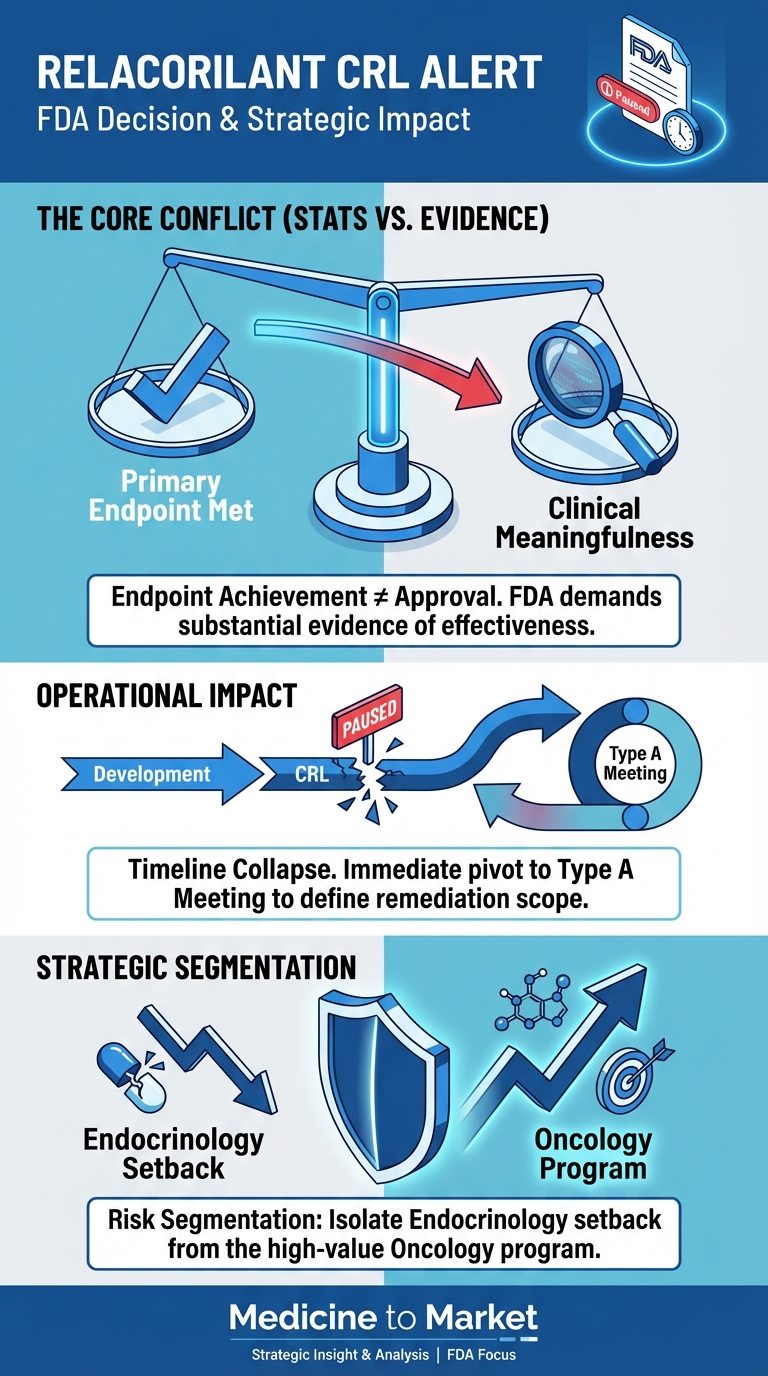
FDA Issues Complete Response Letter (CRL) for Corcept Therapeutics’ Relacorilant: Analysis and Pipeline Impact
What’s Next for Corcept Therapeutics After FDA’s CRL for Relacorilant as a
Patient support, adherence, and advocacy

What’s Next for Corcept Therapeutics After FDA’s CRL for Relacorilant as a

The State of HCP Engagement 2026: Separating Vendor Hype from Commercial Reality

In today’s healthcare scene, pharma firms work with many groups. Each group

In a world of drug tests, trials must show all lives. Experts

In today’s health field, change hits fast. HCPs learn new things. Georgia

Artificial intelligence (AI) shifts U.S. healthcare. AI touches care, tests, and drug

The pharmaceutical landscape is witnessing significant changes with the recent implementation of

Bayer’s recent Phase 3 clinical trial for its innovative blood-thinner, asundexian, has

The landscape of Alzheimer’s treatment took a notable downturn recently when Johnson

The 340B Drug Pricing Program, designed to provide discounted medications to economically

On November 14, 2025, the U.S. Food and Drug Administration (FDA) made

In a significant shift in the biopharmaceutical landscape, Bristol Myers Squibb and

In a significant move that underscores the evolving landscape of cancer drug

The landscape of cancer treatment is evolving, especially for patients battling acute

In an era where heart disease remains a leading cause of mortality

In a groundbreaking clinical trial conducted in China, the innovative drug ivonescimab

In a significant move in healthcare policy, President Donald Trump has brokered

Supernus Pharmaceuticals has recently hit the brakes on new deliveries of its

In the ever-evolving landscape of pharmaceuticals, competition can often be perceived as

The introduction of TrumpRx.gov marks a pivotal moment in the healthcare industry,