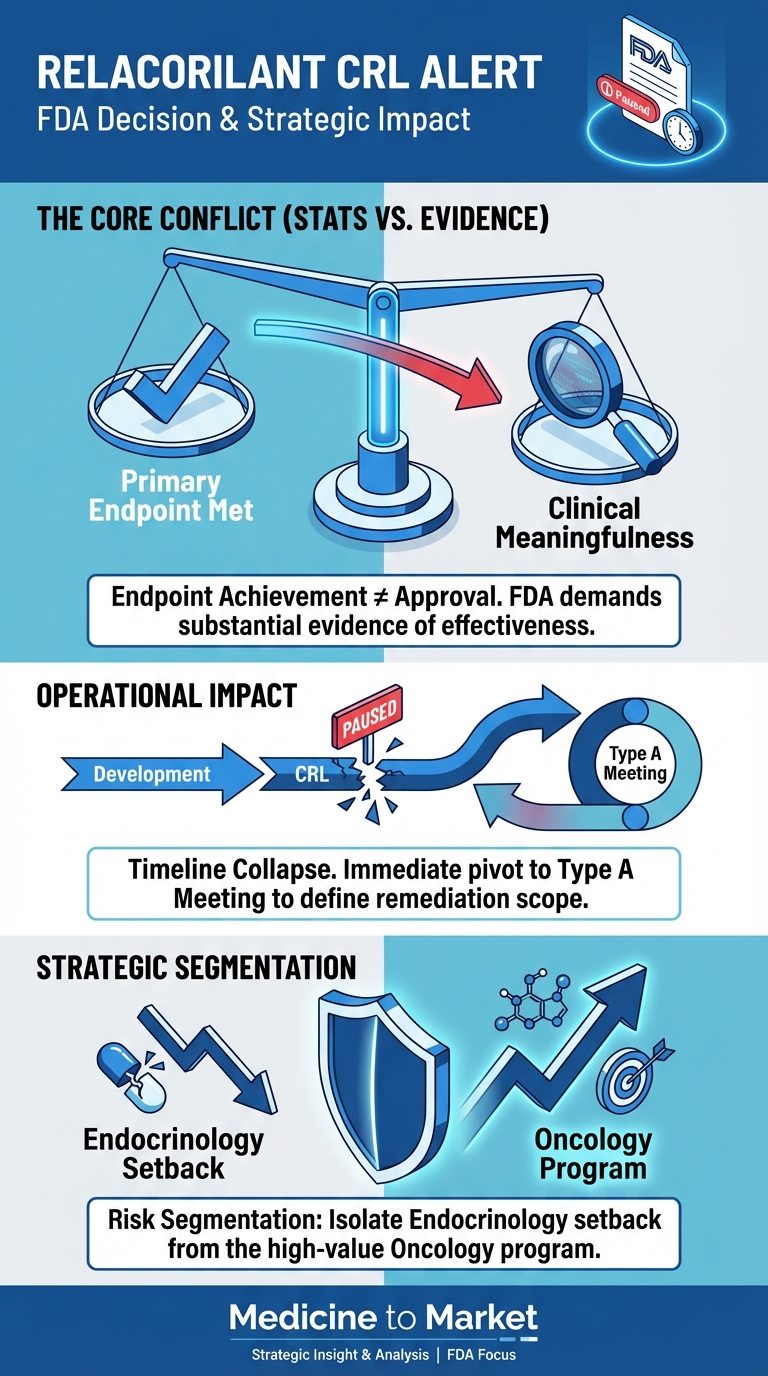
FDA Issues Complete Response Letter (CRL) for Corcept Therapeutics’ Relacorilant: Analysis and Pipeline Impact
What’s Next for Corcept Therapeutics After FDA’s CRL for Relacorilant as a
FDA, EMA, and regulatory affairs

What’s Next for Corcept Therapeutics After FDA’s CRL for Relacorilant as a

The State of HCP Engagement 2026: Separating Vendor Hype from Commercial Reality

In today’s fast pharma scene, brands act. Customer talk must hit, feel

Artificial intelligence (AI) shifts U.S. healthcare. AI touches care, tests, and drug

The recent appointment of Richard Pazdur as the head of the FDA’s

Navigating the complex landscape of pharmaceutical development requires a deep understanding of

In the intricate world of pharmaceutical regulation, the intersection of politics and

November 21, 2025 HEADLINE FINDINGS: Alkermes escalates bidding war for Avadel Pharmaceuticals to

Redemplo (plozasiran) cleared for familial chylomicronemia syndrome at $60,000 annual price—ten times

Biotech prepares FDA submission despite failing key pain crisis endpoint; shares plummet

Agios Pharmaceuticals recently faced a substantial decline in its stock value following

BIOPHARMA EXECUTIVE INTELLIGENCE BRIEFING November 19, 2025 Headline Findings: Merck has announced

On November 18, 2025, Arrowhead Pharmaceuticals made a historic move by receiving

In a bold move aimed at revitalizing American pharmaceutical research, FDA Commissioner

The recent announcement from the White House about a new trade deal,

On November 14, 2025, the U.S. Food and Drug Administration (FDA) made

The landscape of cancer treatment is evolving, especially for patients battling acute

In a significant move, President Donald Trump recently signed a short-term government

Malaria remains one of the world’s deadliest infectious diseases, with millions of

In recent weeks, the U.S. Food and Drug Administration (FDA) has witnessed